Use Chemistry to Hatch a Dinosaur Egg!

Got a dinosaur enthusiast at your place? Make a dinosaur egg they can hatch with a chemical reaction!
You Need:
A packet of Sodium Bicarbonate (baking soda, from the supermarket)
Some food colouring
Mixing Bowl
Water
Gloves
Small dinosaur figurines (or other animals that hatch out of eggs, such as lizards or birds)
Baking tray with a sheet of baking paper or foil to line it
Vinegar in a small cup or bowl
Pipette (eye dropper) or small spoon
What To Do:
Pour the sodium bicarbonate into the mixing bowl. Put on the gloves, then add a little water and a few drops of food colouring. Mix with a spoon or your hands to form a mouldable paste, adding more water if necessary.
Take a handful of the paste and mould it into a small bowl that you can place a figurine inside. Place more paste on top of the figurine and mould into an egg shape, making sure the figurine is totally covered. Place it on the lined baking tray. Use the remaining bicarbonate paste to make one or two more eggs. Place all the eggs on the tray, then place the tray into the freezer for a few hours so the eggs can harden.
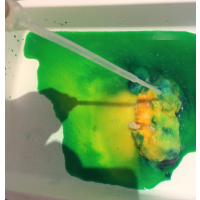
When your eggs are nice and hard, take them out of the freezer and place in a plastic bowl or tray. Give the vinegar to your dinosaur fan and show them how to use the pipette to squirt vinegar onto the egg. Use a spoon if you do not have a pipette. Once the dinosaur is ‘hatched’, rinse it under water before handling it.
NOTE: Small children will need to be shown explicitly how to use a pipette to transfer the vinegar.
Why Is It So?
The vinegar dissolves the sodium bicarbonate (this acid-base reaction is used is many junior science activities, such as making volcanoes, as it is safe and cheap). The ‘fizzing’ that you can see and hear is the production of carbon dioxide gas during the reaction. Chemical reactions are an ‘irreversible change’ – we can not get the original starting materials back again. This activity provides a good contrast with our other dinosaur egg activity, which uses a physical change.